Rivets and welds are by far the most popular choices for fastening handles onto lids and cookware. Screw and body-extension handles are other common ways of affixing handles to cookware. I would recommend welded handles whenever it’s within your budget. I wouldn’t reject a pan solely on the basis of having rivets, though, especially if there are no alternatives that fit your budget.
Cookware Material: Is Enamel safe? Is Ceramic safe? A look at Enamel Glass and Ceramic (Thermolon, Corning Visions, Porcelain, Stoneware, etc.)
Cooking surface: 2/5 to 4/5 (Poor to Good depending on the exact formulation; numerous reports of stickiness even for allegedly nonstick ceramics; on the other hand, enamel/glass/ceramics are mostly non-reactive with food)
Conductive layer: 1/5 Very Poor (enamel/glass/ceramic is a terrible heat conductor by itself)
External surface: 3/5 Fair (chipping is common, but careful usage and buying only thick, high-quality EGC can go a long way)
Examples: Fontignac, Le Creuset, Lodge, Staub, Pyrex, Corning Visions
Health safety: 5/5 Excellent (non-reactive)
—–
DESCRIPTION AND COMPOSITION

Glass is non-crystalline material like Pyrex, made from sand/stone/soil. Glass is hard, brittle, and impermeable.
Enamel is powdered, melted glass used to coat something else, such as enamel coating over cast iron.
Ceramic is a general term which encompasses stoneware, porcelain, and earthenware. Ceramics are hard, brittle, and impermeable like glass. Stoneware and porcelain are examples of clays fired to high temperatures.
Stoneware is fired clay and is opaque and more or less nonporous and nonabsorbent (2-5 percent absorption). Like glass, it is hard, brittle, and impermeable, so water and air can’t fully penetrate.
Porcelain is a special type of high-grade stoneware that uses kaolin clay. Porcelain is more durable than other stoneware and is nonporous, translucent, and especially nonabsorbent (0-1 percent).
Earthenware is not fired as high as stoneware. Earthenware is not impermeable, so liquids can get through eventually. It should not be used as general-purpose cookware. We won’t be considering it further. [Read more…]
Cookware Material: Copper (Cuivre)
Cooking surface: 1/5 Very Poor (large amounts of copper is toxic to ingest)
Conductive layer: 5/5 Excellent (the best affordable thermal conductor)
External surface: 3/5 Good (softer than stainless but still harder than aluminum; oxidizes rapidly)
Examples: Mauviel, Matfer Bourgeat
Health safety: 1/5 Very Poor (toxic in high doses, which is why it’s usually lined with another material on the cooking surface)
—–

DESCRIPTION AND COMPOSITION
Many people enjoy copper cookware’s shiny, lustrous, pink-orange looks. Copper cookware has historically been the pinnacle of affordable (read: non-silver) cookware, used by such famous cooks as Julia Child. But copper, also known as “cuivre” in French, is more than just a pretty face; it’s practical for cooking as well. Among all metals at room and cooking temperatures, copper is the second-best heat conductor, barely edged out by silver which is less than 10% more conductive (the exact percentage depends on metal purity). Copper is more thermally conductive than gold as well, not that many people would be foolish enough to make cookware out of gold.1 Indeed, 1 mm of copper thickness is worth ~2.3 mm of aluminum alloy thickness in terms of heat-spreading power, at least in theory. (In practice the ratio is more like 2:1.) [Read more…]
Cookware Even Heating Testing Methodologies
COOKWARE EVEN HEATING TESTING METHODOLOGY
I went through several iterations of testing methodologies before settling on two that I thought were realistic and representative of how people use their cookware. I’m also planning on a couple of upgrades and additions to these tests that I thought up on my own, but if you have suggestions on cookware to test or how to make testing methodology even better, let me know.
OLD Electric (Induction, Coil, Radiant, Halogen) Testing
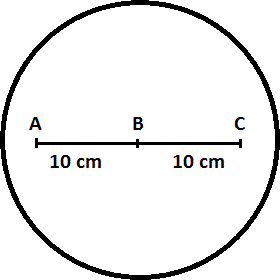 1)” width=”280″ height=”280″ /> Diagram of Pan Bottom (Point B = 350 degrees F 2)
1)” width=”280″ height=”280″ /> Diagram of Pan Bottom (Point B = 350 degrees F 2)
Bottom Line: the lower the temperature delta (difference), the better, and ideally cookware would have a 10 cm temperature delta of zero. That is, ideally the entire cookware surface should be the same temperature.
Cookware Even Heating Rankings (Butane, Propane, Natural Gas, etc.)
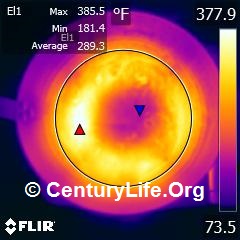
For electric coil/induction/radiant/halogen rankings, please see Even Heating Rankings (Induction and Electric). For more details on testing methodology, see here.
EVERY COMPANY CLAIMS THAT THEIR COOKWARE IS EVEN-HEATING, BUT IS THAT TRUE?
The job of stovetop cookware is a) to smooth out the uneven heat coming from the burner underneath so that the entire cooking surface of the cookware is the same temperature; and b) to keep your food in the Maillard reaction temperature zone–not too hot and not too cold. If you have too much of an imbalance in temperatures, you wind up with hot or cold spots that can undermine your dish by leaving some food overcooked and some food undercooked. You may even scorch carcinogens into your food or produce carcinogenic smoke, if some hotspots grow hotter than the oil’s smoke point while you are waiting for non-hotspots to catch up in temperature. (Hotspotting is particularly troublesome on nonstick pans because you can overheat the hotspots when trying to bring up the temperature of the colder spots, and overheating PTFE/Teflon will permanently damage it and cause offgas.) If you’ve ever cooked fish where part of a fillet got overcooked while the rest was undercooked or made rice and had some undercooked while the rest burned, then you’ve already experienced the joy of uneven heating.
The larger the diameter of your cookware bottom relative to the diameter of the flame or heating element or induction coil, the bigger the uneven heating problem can be, since heat has to travel a longer distance to reach the sides.1
Even Heating Cookware: Why Even Cooking Matters And What We Can Do About It
WHY DO WE WANT EVEN HEATING AND WHAT CAN WE DO ABOUT IT?
The main job of the stovetop cookware is to smooth out the uneven heat coming from the burner underneath so that the cooking surface of the cookware is the same temperature. If you have too much of an imbalance in temperatures, you wind up with hot or cold spots that can undermine your dish and your health by leaving some food overcooked and some food undercooked. You may even scorch oil and produce carcinogenic smoke, if some hotspots grow hotter than the oil’s smoke point. If you’ve ever cooked fish where part of a fillet got overcooked while the rest was undercooked; if you’ve ever made rice and had some undercooked while the rest burned; if you’ve ever burned part of a strip of bacon while waiting for the ends of the strips to be done, then you’ve already experienced the joy of uneven heating.
A second job of cookware is to hold that heat so as to maximize Maillard reactions. These reactions take place at higher temperatures. Thus if you have a flimsy pan and tossing in a steak crashes the temperature, that will mean more steaming/boiling of your food and lower Maillard reactions, resulting in less-tasty food.